Flu vaccination 2018/19 €“ SOPs and guidance
In NPA Essential
Follow this topic
Bookmark
Record learning outcomes
The flu season for 2018/19 is now here. To support pharmacy teams in delivering both NHS and private vaccination services, the NPA launched a suite of new resources containing a range of SOPs and guidance. These will assist teams in preparing for, and delivering high quality vaccination services.
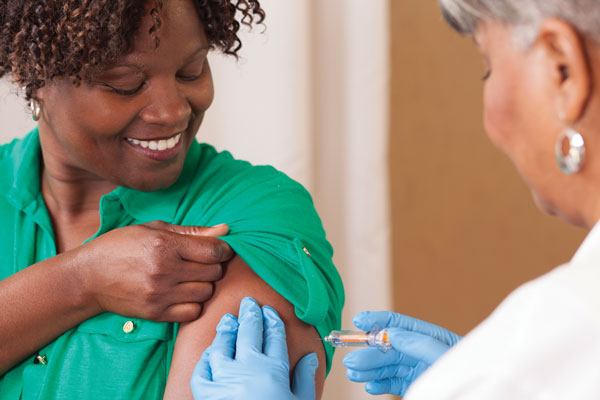
The resources include:
- €Flu vaccination general guidance 2018-19€
- €Flu vaccination guidance (England) 2018-19€
- €National flu vaccination Advanced Service (England) 2018-19 SOP€
- €Flu vaccination guidance (Wales) 2018-19€
- €Pharmacy team guide: recognising flu€
- €Administration of adrenaline SOP€
- €Hand hygiene SOP€
- €Maintaining cold-chain SOP€
- €Needlestick injury and biohazard spillage SOP€
- €Delegation of responsibilities€
- €Pharmacy team declaration€
- €Chaperone policy: FAQs€
- €Chaperone policy: guidance€
- €Chaperone policy: patient notice€
- €Chaperone policy template€
- €Cold-chain products: date-checking matrix€
- €Cold-chain products: drug alert log€
The standard operating procedures (SOPs) listed above should be used in conjunction with the €Delegation of responsibilities€ and €Pharmacy team declaration€ resources.
SOP Guidance:
- IMPORTANT: The RP must be signed in to provide an Advanced Service.
- Refer to the €delegation of responsibilities€ notes to ensure that for each action/activity to be delegated, it takes into account the competency of the relevant team member to carry out that action/activity
Preparing for the service
- Download and print the NHS Community pharmacy influenza vaccination Advanced Service Patient Group Direction (PGD) and service specification; the PGD must be signed by the pharmacist (who has been received appropriate training and is authorised to use the PGD), and the Authorising Manager (this would be an individual with the relevant level of authority)
- Ensure relevant members of the pharmacy team have undertaken appropriate vaccination training and completed the Declaration of Competence (DoC); pharmacists must review their DoC every two years
- Each staff member undertaking the service should consider receiving vaccination against hepatitis B €“ refer to Appendix 2
- Off-site vaccinations for patients at long- stay care home or residential facilities can only be conducted with prior approval from NHS England; refer to the service specification for more information.
Cold chain
- Vaccines are to be stored as instructed by the manufacturer
- Maximum-minimum refrigerator temperatures must be recorded daily when the pharmacy is open
- Refer to NPA 'maintaining cold chain' SOP for further information.
Delivering the service €“ ensure:
- All necessary equipment, including a sharps bin, is readily available and easily accessible
- The consultation room is clean, presentable, and meets the requirements specified in the service specification
- Adrenaline ampoules for injection 1:1000 are immediately available, with appropriate sterile needles and syringe
- Ensure other members of the pharmacy team are available in case of an emergency, for example, to call for an ambulance if the patient suffers an adverse event
- That patient confidentiality and privacy are maintained at all times
Additionally
- The consent form must be signed by the patient before administration; this form also permits the sharing of information with the patient's GP and NHS England for the purpose of post-payment verification
- The patient questionnaire can be completed electronically or on paper; an NHS Flu Data collection portal will be available for pharmacies to enter questionnaire responses for analysis
- For further information regarding immunisation procedures, refer to NPA flu resources
- Refer to relevant NPA SOPs; hand hygiene, cold chain and needlestick injury.
Record keeping
- NHS England state €records of patient vaccinations should be kept for an appropriate length of time€ for payment purposes and this should be for a minimum of two years
- It will be a decision for the pharmacy contractor to decide how long beyond two years the information should be stored but it is recommended that all patient consent forms relating to any Advanced or Enhanced Service are retained for six complete tax years
- For further information related to record keeping, refer to Appendix 1, the flu PGD, service specification for 2018/19 and NPA €Record keeping: guidance€.
All the above resources are available now for NPA members to download from the NPA website. For further information please contact the NPA Pharmacy team on 01727 891800 or email at: pharmacyservices@npa.co.uk.
